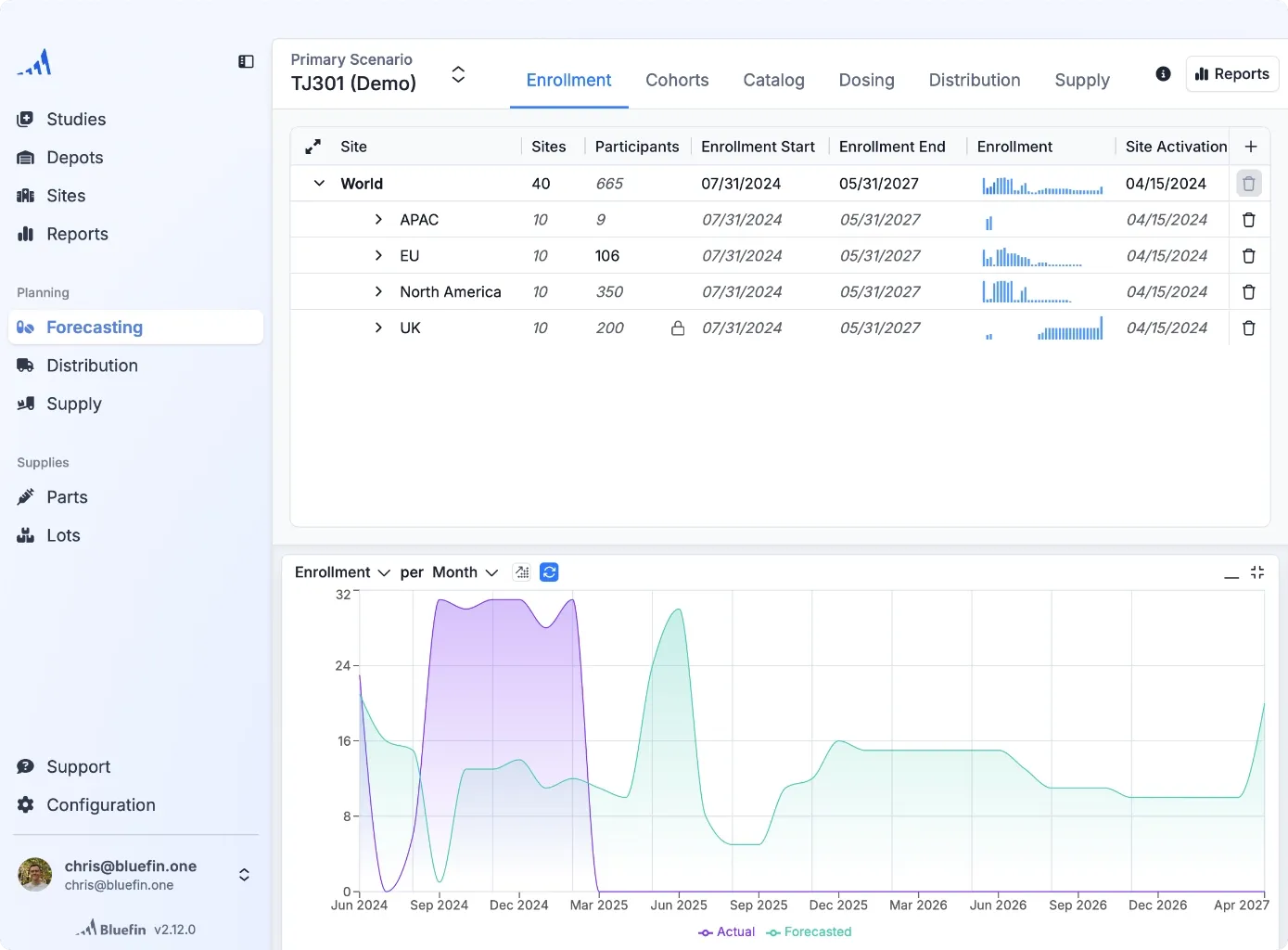
Structured, consistent forecasting across your team and over time
Reduce overage and risk with better forecasts and no formula errors
Automate supply planning using AI and advanced algorithms
Creating a pack/label plan in Bluefin takes minutes. Before we had Bluefin it would take me hours. When we're working on a deadline like a meeting a few days out, this is the difference between success and failure.Clinical Supply ManagerTop-50 Pharmaceutical Company
Forecast any study, fast.
Right amount, right time.
Automated, optimal shipment planning.
Secure and compliant platform.
Drive strategic impact with advanced clinical supply forecasting
Let's discuss what Bluefin can do for your trial.